Is An Arthritis Drug The Answer To Alopecia?
11th July 2014
Most people wouldn’t think to link arthritis to alopecia – until now. An American dermatologist has reportedly restored the hair of an alopecia areata sufferer by administering a drug more commonly used to treat rheumatoid arthritis.
As many of you know, alopecia areata is an ailment that occurs when the body’s immune system attacks its own hair follicles. Estimated to affect around 1.7 per cent of the UK population, the condition is known for its sudden and random onset. Although hair loss of any type doesn’t have any affect on a person’s physical health, it can leave a sufferer’s confidence and self-esteem in tatters.
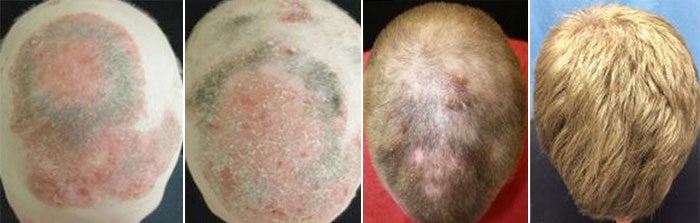
Dr Brett King, a scientist at Yale University in Connecticut, claims to have cured his patient’s alopecia areata by prescribing the drug tofacitinib citrate, which is made under the brand name of Xeljanz. The drug is currently approved by the US Food and Drug Administration organisation (FDA) for the treatment of the autoimmune disease, rheumatoid arthritis.
Prior to the treatment, Dr King’s 25-year-old patient was almost totally hairless, having suffered from alopecia for the past seven years. Following eight months of treatment – where he received 10mg of tofacitinib citrate every day for two months followed by 15mg per day for three months – the patient had achieved a remarkable re-growth of hair all over his body.
As well as alopecia areata, the patient also suffered from another autoimmune disease, plaque psoriasis. The drug has been tested in mice with alopecia through Prof. Angela Christiano at Colombia University, with the results widely known. Dr King and his team decided to trial the drug as a combined treatment for both ailments, believing that it would work by turning the immune system off.
It’s great to see that scientists are working towards new and interesting treatments to combat alopecia, but I’m wary of the use of Xeljanz to treat a condition that has no affect on a person’s physical health, due to the drug’s side effects. Xeljanz is widely known to increase the risk of cancer and gastro-intestinal perforations, and was consequently rejected for licensing in Europe in 2013. While this therefore isn’t likely to pose a solution to the millions of alopecia areata sufferers in the world, it’s very encouraging to see scientists look for new ways to treat it.
Another important thing to keep in mind is that alopecia areata is an extremely unpredictable condition and the results Dr King’s patient experienced could actually have been a placebo effect, as opposed to a medical breakthrough. Unfortunately, any success with this drug is not going to apply to male or female hereditary type hair loss as these are completely unrelated conditions.


